Controlled Drugs: Reminder on Safe Custody and Record-Keeping Expectations
WHY THIS MATTERS NOW
Controlled drugs remain one of the highest-risk areas of pharmacy practice, attracting sustained regulatory scrutiny due to their potential for misuse, diversion and patient harm. This week, pharmacy teams are being reminded of established but critical expectations around safe custody and accurate record-keeping, following renewed emphasis from regulators and NHS bodies.
CORE STORY
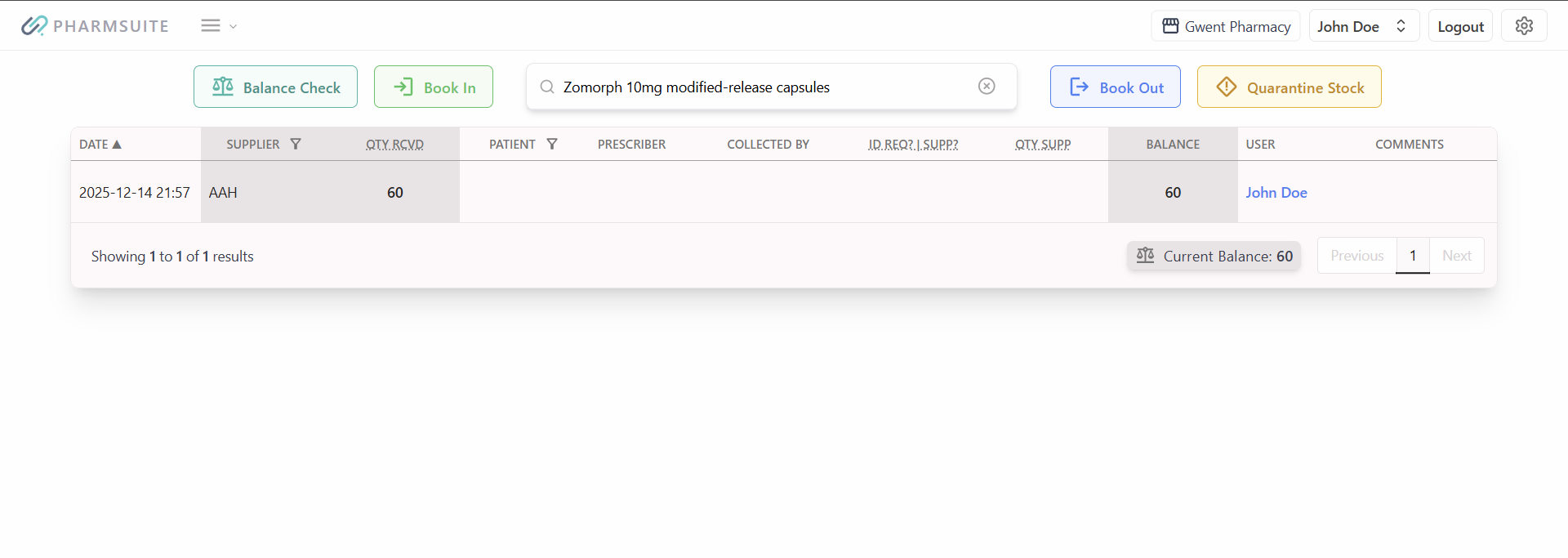
Recent regulatory communications and inspection feedback continue to highlight controlled drugs (CDs) as a recurring area of concern in pharmacy settings. Issues most commonly identified include incomplete or delayed CD register entries, discrepancies between physical stock and records, and weaknesses in safe custody arrangements.
While no new legislation has been introduced, regulators have reinforced that existing Misuse of Drugs Regulations must be followed rigorously. This includes maintaining an accurate, contemporaneous CD register, ensuring secure storage that meets legal standards, and promptly investigating and documenting any discrepancies.
These expectations apply across community and hospital pharmacy settings in England, Wales and Scotland.
PRACTICAL IMPLICATIONS FOR PHARMACY
Pharmacy teams should take time to review CD standard operating procedures and ensure all staff involved in handling controlled drugs are trained and competent.
Regular balance checks, clear handover processes and timely register entries remain essential safeguards. Where errors or discrepancies occur, these should be recorded, investigated and escalated in line with local governance procedures.
Locum pharmacists should also be supported with clear guidance on local CD processes to reduce the risk of documentation gaps.
WIDER CONTEXT
Controlled drugs governance continues to be a focal point for inspections because failures can have serious consequences for patient safety and public trust. As workloads increase, regulators are increasingly attentive to whether systems remain robust under pressure.
CLOSING INSIGHT
Controlled drugs management is an area where small lapses can have significant consequences. Consistent processes, clear documentation and regular checks remain the cornerstone of safe and compliant practice.